The successful drop in reported leprosy cases worldwide — from 1.15 million in 1997 to 228,474 in 2010 — is attributed largely to the antimicrobial multidrug therapy (MDT) developed in 1981. Listed as a neglected tropical disease by the World Health Organization (WHO), leprosy is an infectious disease of the skin and nerves caused by Mycobacterium leprae. Resistance to antimicrobial drugs evolves out of genetic mutations in the pathogen. The MDT, consisting of dapsone, rifampicin, and clofazimine, was developed in response to dapsone resistance in monotherapy during the 1960s. While clofazimine resistance has not been described, resistance to rifampicin — the backbone of MDT — has appeared. Controlling leprosy transmission requires not only early detection, but routine surveillance for mutations in the drug target genes — folP1, rpoB, gyrA — of M. leprae before, during, and after the course of treatment.
Dr Varalakshmi (Vara) Vissa, a member of the leprosy research group at Colorado State University (CSU), has developed molecular epidemiological methods to exploit the M. leprae genome for clinical applications, such as the identification of transmission clusters, diagnostics, monitoring of treatment efficacy, evolution of drug resistance, and possible discovery of drug and vaccine targets. Vissa’s lab has developed novel real-time PCR high resolution melt (HRM) analysis assays to quickly, simply, and inexpensively identify the presence or absence of the specific mutations known to confer drug resistance.

Assistant Professor
Department of Microbiology, Immunology, and Pathology
Colorado State University
“I wanted to get away from sequencing and harness the data right at the stage of PCR itself,” says Vissa, associate professor of microbiology, immunology, and pathology at CSU. “You can use one plate, one machine, and extract information based on the difference in melting temperature of the amplicons — that’s the power of HRM.”
Traditionally, target loci were amplified through PCR followed by DNA sequencing to detect mutations. However, PCR DNA sequencing is time and labor intensive for routine drug surveillance, and for the single nucleotide polymorphism (SNP) strain typing used to trace leprosy transmission. To find one or two mutations the entire fragment must be sequenced, which is typically performed off-site. Also, a mixture of wild type and mutant bases is difficult to delineate above background peaks in sequence chromatograms.
Although the Vissa lab used surrogate PCR-restriction fragment length polymorphism (RFLP) assays previously, this method proved unsuitable for the current project. The drug mutations lie in one or several codons located within short stretches of DNA in each target gene, referred to as the drug resistance determining regions (DRDRs). DRDR assays are not suitable for PCR-RFLP as there are different mutations at one or more positions and no convenient restriction enzyme sites to overlap these regions.
With assistance from Bio-Rad field specialists, Vissa developed new HRM assays for 1) surveying global drug resistance (the DRDR assays), and 2) tracing leprosy transmission (the SNP assays). In HRM analysis, amplified DNA is denatured in precise temperature increments to produce melt curves with features that are dependent upon nucleotide sequence. Curves with similar shapes are derived from the same DNA sequence and can be clustered together, allowing researchers to analyze genetic variants post-PCR. This technique proved inexpensive and convenient for the preliminary screening of DNAs and rapid classification of clinical strains into wild type or variant clusters, which carry some of the known mutations in the DRDRs. For folP1 (dapsone resistance) and rpoB (rifampicin resistance), three different proven drug resistance mutations were tested, and could be separated from wild type. DNA amplification prior to HRM analysis was used for quantitative PCR (qPCR) to estimate the starting template amount in clinical DNA samples attesting to the utility of the HRM workflow.
HRM proved equally informative for the SNP typing assays, offering rapid, robust classification. SNP assays are typically the most challenging due to the fine melt profile differences resulting from small sequence variations. The novel SNP typing assays proved suitable for comparing M. leprae strains on global and national levels.
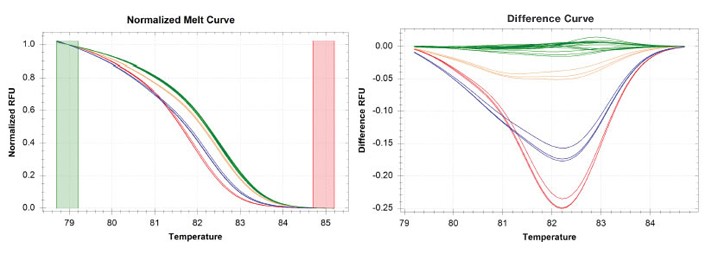
gyrA gene panel depicting genotypes of the mutant strains against the wild type NHDP63 strain. gyrA tests for fluoroquinolone resistance. Fluoroquinolones may be required when rifampicin resistance is encountered or other contra-indications are present. Wild type is shown in green and the mutant strains are shown in orange, blue, and red.
With Bio-Rad’s assistance, the lab was able to optimize the PCR reagents and experimental HRM conditions to capture a double mutation in the folP1 DRDR (dapsone resistance) with a negligible melting temperature (Tm) shift, demonstrating the power of Bio-Rad’s CFX96™ real-time PCR detection system’s optics and the efficacy of the precision melt supermix for resolving small thermal profile differences.
“Originally Dr Vissa’s lab was using a non-HRM-formulated supermix and having trouble differentiating a double point mutation to resolve the wild type from mutant in a 78 bp amplicon,” says Paul Streng, real-time PCR development specialist at Bio-Rad. “We introduced them to the precision melt supermix and focused on a 52 bp amplicon, achieving success. At the time, the mix was not yet commercially available.”
Overall the HRM assays saved time and money for the lab since there was no need for additional post-PCR procedures. For each target amplicon, the assay costs less than $3 per sample. In comparison, a single sequencing reaction per target for just one direction costs $7–10 at a subsidized rate.
Leprosy Transmission
Leprosy transmission continues because of clinical resistance in infected populations and the disease’s long incubation time.
“With a long incubation there’s the risk of continued transmission,” says Vissa. “We don’t know how many reservoirs of people there are who haven’t been diagnosed and are pre-symptomatic carriers. The problem is how to identify these segments of the population. Only when you lose the infective population can you eliminate the disease.”
Future Research
In conjunction with HRM analysis, Vissa is hoping to construct a synthetic library across all the DRDRs. Eventually her lab would like to use additional methods to investigate more targets in the M. leprae genome through multiplexing and next generation sequencing. The aim is to more quickly identify and interrupt leprosy transmission networks.
“We don’t want to miss someone,” says Vissa. “We want to identify all the DRDR mutations. Our work is just beginning. We demonstrated that in one sweep we could capture quite a lot of information just with the HRM itself. Now we’re looking to secure funding to continue collaborating with colleagues and include next generation sequencing work as well.”
Centers around the world where the Vissa lab collaborates on leprosy research.
In the meantime Vissa’s novel HRM assays, developed with the help of Bio-Rad specialists, offer researchers an expedited molecular technique to keep vigil on drug sensitivity patterns to sustain declining trends of leprosy in endemic countries, and ultimately bolster the success of antimicrobial therapies like MDT.
Vissa would like to acknowledge the receipt of an American Recovery and Reinvestment Act (ARRA) grant, which provided funds for exploring emerging technologies to accelerate her research.
The complete information on Vissa’s work on HRM optimization for detecting drug resistance mutations in leprosy can be found in the following research paper:



