
Ligand Immobilization in Protein Interaction Studies — An Unattended Amine Coupling Protocol with Automatic Coinjection Activation

Novel Liposome-Capture Surface Chemistries to Analyze Drug-Lipid Interaction Using the ProteOn™ XPR36 System
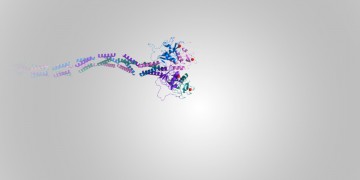
Building New Anti-HIV Molecules with Bio-Rad’s ProteOn™ XPR36 System
Efficient SPR-Based Fragment Screening and Small Molecule Affinity Analysis Using Bio-Rad’s ProteOn™ XPR36 System
Guide to SPR Data Processing on the ProteOn™ XPR36 System
Guide to SPR Data Analysis on the ProteOn™ XPR36 System

Options for Dataset Export Using the ProteOn Manager™ Software

A Novel Biotinylated Ligand-Capture Method with Surface Regeneration Capability for Label-Free Biomolecular Interaction Analysis

Immobilization of Active Kinases for Small Molecule Inhibition Studies


