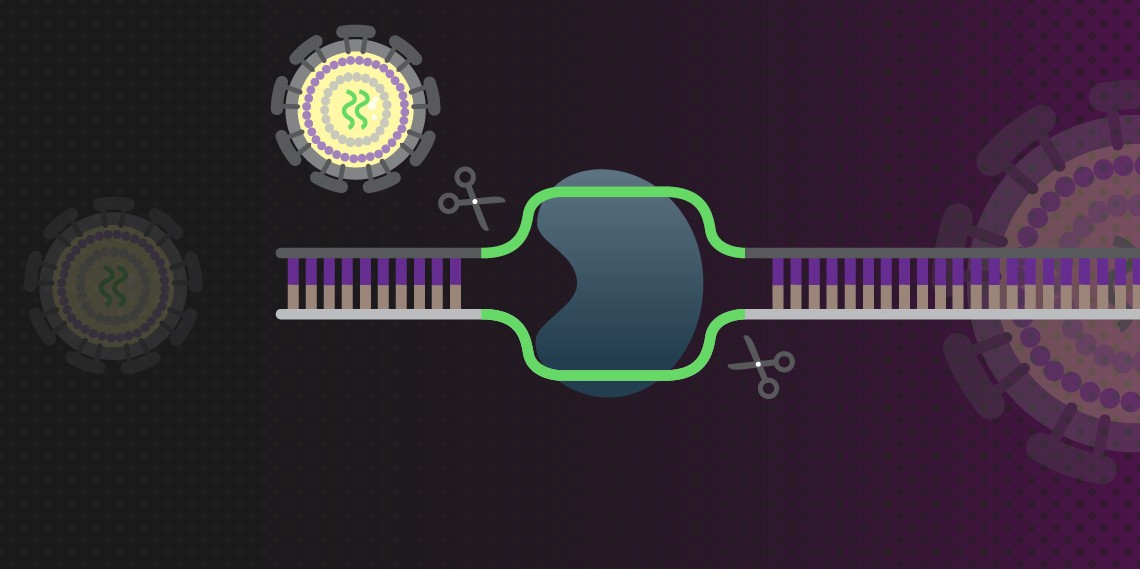
The advent of CRISPR as a gene editing tool appears to have revolutionized drug discovery and development in a very short time. The CRISPR-Cas9 system uses short RNAs to direct the Cas9 nuclease to target specific DNA sequences. Because it uses RNA rather than a protein to target nuclease activity, CRISPR-Cas9 can be easily and accurately retargeted by synthesizing a new guide RNA (gRNA).
The gRNA also makes this technique amenable to high-order multiplexing, that is, large-scale screens using hundreds or thousands of gRNAs in a single experiment. Consequently, complex animal models that once took years to develop are now completed in just a few months and at a fraction of the cost.
CRISPR’s ease of use, early successes, and relatively low associated costs have enhanced its use in many areas of research. For a more comprehensive overview of the potential capacity for CRISPR, read: Erasing Disease: How Gene Editing is Changing Genetic Therapies. For an overview of CRISPR disease models, read: CRISPR: Changing the Pace of BioPharma R&D.
The Relationship between HIV and AIDS
For more than 30 years, HIV has been a leading public health issue and a disease for which there has been an earnest search for a cure. Globally, 37 million people currently have HIV infections. An equal number of lives have been lost since the virus’ formal discovery in 1983. Although there have been significant declines in the annual number of new infections and AIDS-related deaths since 2010, there were still 1.8 million new HIV infections documented in 2016 (Terbush 2017).
Modern treatments, mostly antiretroviral drug therapies (ARTs) that target replicating viruses, are largely the cause of the decline in the death rate. However, these treatments are not a cure and must be taken daily for the remainder of the patient’s life. If ARTs are discontinued, latent virus can be reactivated and will eventually lead to AIDS.
During infection, the HIV virus deposits a copy of itself into the cell’s genetic material, making the infection a permanent part of the genetics of the cell. From this integrated viral DNA (the provirus), copies of HIV can be made. If left untreated, HIV eventually infects and destroys T cells leading to AIDS, which destroys the body’s ability to fight infection (Wang and Cannon 2016). Although ARTs can help postpone AIDS, a growing issue with these drug-based therapies is the side effect of neurocognitive disorders (Ebina et al. 2016). In addition, ARTs and their associated costs are an ongoing burden for many countries’ healthcare systems.
Gene Editing Technologies Targeting HIV Infection
HIV/AIDS has been called one of the most uniquely suited diseases for developing gene editing applications. Zinc finger nucleases (ZFNs) are currently being studied in phase I and II clinical trials evaluating co-receptor CC chemokine receptor 5 (CCR5) disruptions in T cells and their in vivo precursors, hematopoietic stem and progenitor cells (HSPCs). Preventing the expression of the CCR5 gene represents the first potential application of targeted nucleases in humans since disruption is much easier than gene editing. This gene is an attractive disruption target because as a nonessential human gene its absence provides potent anti-HIV protection. While this seems promising, ZFNs have been difficult to program and a major concern is the possibility for off-target genomic modifications. Additionally, in almost 50% of patients CXCR4-tropic viruses, a variant of CCR5, emerge in the later stages of HIV infection. Disruption of CXCR4 is also being explored; however this approach is limited due to the essential role it plays in the immune response (Wang and Cannon, 2016).
CRISPR-Cas9 has been used to either mutate or cut out the provirus. In the first instance, the gRNA makes a cut through the targeted DNA so that when the cell tries to repair the damage, imperfections or mutations are introduced that disable the effected genes. This method also carries the risk of off-target effects but these have been shown to be dependent upon the targeting sequence. An alternative is to use a double nickase strategy, which generates breaks in a single strand of DNA at specific loci, usually the 3′ or 5′ overhangs on opposite strands. This method lowers the risk of off-target mutations (Kaminski et al. 2016).
Recently, researchers at Temple University and University of Pittsburgh demonstrated that they could excise HIV-1 from the genomes in three animal models, including a humanized mouse model in which mice were infected with HIV after being transplanted with human immune cells (Yin et al. 2017). In this study, they showed that their gene editing model was effective not only in acute infection but also in chronic and latent infection. They utilized a recombinant adeno-associated viral (rAAV) vector delivery system in order to influence a broader range of cell targets and abolish HIV-1 mutational escape. Their next step will be to repeat this study in primates in which HIV actually induces infection and to further show the absence of latently infected T cells hiding in inaccessible organ niches such as brain cells.
Climbing the Last Mountain
There are no efficient methods available to reliably deliver nucleases to HIV target cells in humans, especially to the latent reservoir of virus that exists in patients on ARTs. Although CRISPR-Cas9 based treatments have been shown to work in the laboratory, there are still a number of barriers to overcome before they can be used on patients. Therapies will need to be personalized for each patient’s particular type of HIV-1 virus. Improvements in delivery are also required so that the majority of circulating T cells is targeted. If these potential cures are shown to work, they have the capacity to lower the number of people with HIV/AIDS-related health issues.
References
Terbush A (2017). Scientists wield gene editing with CRISPR in the fight against HIV. berkeleysciencereview.com/scientists-wield-gene-editing-crispr-fight-hiv/, accessed January 31, 2018.
Wang CX and Cannon PM (2016). The clinical applications of genome editing in HIV. Blood 127, 2,546–2,552.
Ebina H et al. (2016). Perspectives of genome-editing technologies for HIV therapy. Current HIV Res 14, 2–8.
Kaminski R et al. (2016). Elimination of HIV-1 genomes from human T-lymphoid cells by CRISPR-Cas9 gene editing. Sci Rep 6, 22555.
Yin C et al. (2017). In vivo excision of HIV-1 provirus by saCas9 and multiplex single-guide RNAs in animal models. Mol Ther 25, 1,168–1,186.
Bio-Rad is a trademark of Bio-Rad Laboratories, Inc.