The ChemiDoc Touch Imaging System is Bio-Rad’s new digital imaging solution for gel and western blot analysis. Designed for efficiency and ease of use, it integrates a touch-screen interface with Image Lab™ Touch Software to give users a fast workflow for acquiring and analyzing publication-quality image data. As a part of Bio-Rad’s V3 Western Workflow, the ChemiDoc Touch Imaging System enables a western blotting protocol improved with multiple validation steps, streamlined completion times, and quantitative results using stain-free gel chemistry.
We gave this new imaging system to several researchers to test. Each one started the process as a user of X-ray film for western blot imaging. Some reported having tried digital imaging in the past, but not being convinced at that time to give up film. Each sat down for an interview regarding how he used the ChemiDoc Touch Imaging System and provided a representative blot image created using the instrument.
Distinguishing the Best Targets for Novel Leukemia Treatments
In this video, Ernesto Diaz-Flores describes using the ChemiDoc Touch Imaging System to distinguish potential therapeutic targets in translational leukemia research, as well as how his interest in photography gives him a special perspective on digital imaging for protein analysis.
In early 2013, Nature Genetics published a study by a group of researchers that provided the first broad characterization of hypodiploid acute lymphoblastic leukemia, a subtype of childhood leukemia that has proven highly resistant to treatment. Along with a detailed genomic profile, this study also indicated several protein pathway alterations that merited further investigation for possible therapeutic targets.
“As a follow-up to that study, I have been pursuing the protein pathways that we found were most deregulated,” says Ernesto Diaz-Flores, a researcher in Dr. Mignon Loh’s lab at the University of California, San Francisco. “We have been focusing on the Bcl-2 family members, which are involved in the survival pathway. There are 12 to 15 members of this family.”
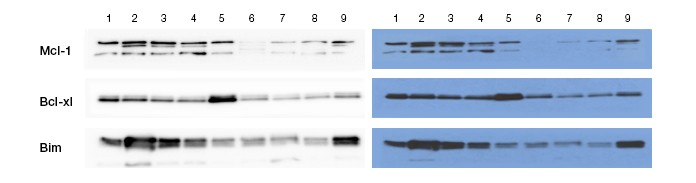
This set of Ernesto Diaz-Flores’s western blot images shows Bcl-2 family proteins in different samples of leukemic cells. Images created using ChemiDoc Touch Imaging System (left panel) and X-ray (right panel) are shown. Diaz-Flores cites the resolution of the ChemiDoc Touch Imaging System for enabling him to visualize very faint bands and to distinguish and quantitate darker bands. This ability provides better understanding of the most relevant targets to investigate for their therapeutic potential in hypodiploid acute lymphoblastic leukemia.
Diaz-Flores and his colleagues found elevated levels of both pro-survival and anti-survival proteins in leukemic cells. These increased levels could indicate a mechanism that enables the leukemic cells to resist treatment. Because these pro- and anti-survival proteins exist in a balance — a balance that was elevated in leukemic cells — Diaz-Flores and his colleagues believe that reducing elevated pro-survival protein levels will upset the balance, inducing cell death in leukemic cells while leaving normal cells untouched. So far, in vitro studies have supported the hypothesis. “Right now we’re moving into in vivo studies to test if these treatments will have the same efficacy as in vitro,” says Diaz-Flores. “If that happens, it would inform a novel therapeutic approach to these high risk leukemias.”
Overcoming challenges to find the best protein targets
Because Diaz-Flores is studying many proteins in multiple cell lines simultaneously, he needs to be able to see fine distinctions between expression levels in order to find the most deregulated — and therefore the most therapeutically relevant — proteins. Discerning and quantitating these differences could prove difficult when using film to image western blots showing expression levels.
“Having a very strong band next to a very faint band, that posed a special challenge,” says Diaz-Flores. “Using film, you had to first try to expose for the dark band, then the faint band. And it was always too much exposure, or not enough. You ended up repeating exposures and wasting time on a technical challenge, rather than a scientific challenge.”
Researcher Ernesto Diaz-Flores loads a western blot membrane into the ChemiDoc Touch Imaging System using Smart Tray Technology™, which recognizes his imaging application, then adjusts the contrast setting on the resulting exposure to optimize the image for his protein of interest.
Diaz-Flores found that the ChemiDoc Touch Imaging System solved this problem by allowing him to quickly optimize exposure for his proteins of interest. “You can indicate your dark band and your faint band. It will expose the best for each band,” he says. This is demonstrated by the image he supplied, which shows expression levels of Bcl-2 family members across nine different cell lines. The ChemiDoc Touch Imaging System better resolved the differences between closely spaced bands, resulting in a clearer measurement of these protein levels.
Quantitating the various proteins’ relative abundance had presented an additional hurdle. “With film you can quantitate, but you cannot quantitate easily,” says Diaz-Flores. “With the ChemiDoc Touch Imaging System, we just look at the levels on the touch screen and quantify them right there. We can see the fold induction or reduction and correlate that to drug response or to other assays, so it’s helped us focus our attention on the most relevant proteins.”
Diaz-Flores also notes that, given the volume of western blots he needs to analyze, streamlining his data acquisition and analysis process with the ChemiDoc Touch Imaging System has helped move his work forward. “Time in science is gold,” Diaz-Flores says. “In a few seconds the machine is able to determine the right exposure and give you a very clean band without extra background noise. Then you can do the analysis right there and export it to your network or a USB drive for a publication or presentation. So the ChemiDoc Touch Imaging System has really simplified the way we analyze and quantify proteins.”
Discerning Subtle Modifications in Signaling in Inflammatory Bowel Disease
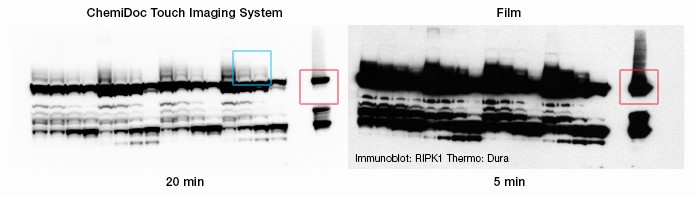
These western blot images from Dr. Ling Shao show RIPK1 (red box), a protein that plays a part in the TNF-α signaling pathway Dr. Shao studies for its role in inflammatory bowel disease (IBD). Oversaturation of bands on an X-ray film (right) had been a problem for Dr. Shao in acquiring blot images, as it had obscured important distinctions between closely spaced bands. With the ChemiDoc Touch Imaging System, Dr. Shao could delineate RIPK1 as well as RIPK1 with one ubiquitin molecule added, with two molecules added, and with three added (the ladder within blue box). What appeared on X-ray film to be a single, overexposed band was revealed by the ChemiDoc Touch Imaging System as containing important data on Dr. Shao’s protein of interest.
Dr. Ling Shao researches inflammatory bowel disease (IBD) at the University of California, San Francisco.* IBD most commonly manifests as either Crohn’s disease or ulcerative colitis, often painful chronic conditions that current scientific thinking believes result from an attack by the body’s immune system against normal commensal flora in the digestive system. Because this inflammatory response begins with a reaction to the body’s microbiome — the collection of microbes that inhabit the body and outnumber “self” cells by ten or more to one — our understanding of IBD has evolved along with our growing understanding of the microbiome’s importance.
“Traditionally, we looked at autoimmune disease as an attack against the self,” says Dr. Shao. “However, much more research is coming out to suggest that to some extent you’re a reflection of your microbiome. So in a way you can think of IBD as an autoimmune disease, but you’ve just got to expand your view of what is you.”
Because of its role in initiating inflammation, the TNF-α pathway is thought to play a key role in IBD. However, Dr. Shao believes that TNF-α has another function in the intestine, to regulate cell death. Both of these processes may be important in the pathogenesis of IBD. Dr. Shao wants to obtain a deeper understanding of the regulation of the TNF-α signaling pathway, which gets its name — “tumor necrosis factor” — from its role in initiating programmed cellular death, or apoptosis, in certain tumor cells. “It is a critical decision” Dr. Shao says, “that the proteins downstream of the TNF receptor help the cell make. Will they respond by amplifying the inflammatory signals or else lead the cell to die through apoptosis?”
In the course of studying the many proteins involved in this signaling network, Dr. Shao performs numerous western blotting experiments. Until recently, he imaged these blots in a darkroom using X-ray film. However, this method has always posed problems to researchers, especially in situations involving a strong signal from closely spaced bands. In such cases, the strong signal often becomes oversaturated, making it impossible to quantitate its abundance and to distinguish it from nearby bands.
Detecting small but significant protein modifications
One protein of interest to Dr. Shao is RIPK1 (receptor-interacting protein kinase 1). This protein plays an important part in the early phases of TNF receptor signaling, especially in helping the cell make the decision to live or die. RIPK1 is modified by ubiquitin as part of this function. Ubiquitin is a small (~9 kD) protein present individually or as a complex, that regulates multiple cellular processes by conjugating with proteins.
Because ubiquitin modifications produce only small changes in RIPK1 sizes, on a western blot image they appear as closely spaced bands. Dr. Shao noted native RIPK1 on the image using a red arrow. In trying to resolve these bands using film, Dr. Shao found that saturation proved to be major issue. “On film, it’s quite easy to overexpose the blot, leading to bands that fuse together,” he says. “Then you might have to re-expose several times to get a usable image, if it’s even possible at all.”
Using the ChemiDoc Touch Imaging System, Dr. Shao was able to delineate the bands that had been obscured by the oversaturated signal on film, which he notes using blue boxes on the image produced using the ChemiDoc Touch Imaging System (left). “On the lanes next to the blue boxes, you can see bands where RIPK1 has been modified by the addition of one, two, and three ubiquitin molecules,” Dr. Shao says. “The bottom band is RIPK1 without any ubiquitin molecules covalently attached (red box). The next one higher has one ubiquitin molecule attached, the next one higher has two, and the highest has three. You can easily distinguish these four bands using the [ChemiDoc Touch Imaging System] whereas with film [left image] these bands are impossible to distinguish.”
Simplifying substrates
This ability results both from the imaging system’s high resolution and its wide dynamic range. Pointing out a related advantage, Dr. Shao emphasizes that to obtain high-quality images, he doesn’t need to test different substrates as he would using film. Instead, he can start imaging with a single substrate and be confident that the imaging system’s dynamic range will accommodate it. “Because the ChemiDoc Touch System has such a great dynamic range, you can just use a stronger substrate to start and you’ll get the different, closely spaced bands that appear on film as a single, dominant band,” he says.
Starting with a substrate with a signal that persists also translates into a workflow advantage. “Unlike with film, I don’t have to continually try to determine what the optimal substrate is for my membrane,” says Dr. Shao. “It ends up being much quicker. If you start with such a substrate, you can avoid having to develop your blot multiple times in order to obtain a publication-quality image. That also decreases the number of substrates you need to have in the lab. We’re going to get rid of most of our substrates and just use one or at most two. So, just pair the [ChemiDoc Touch Imaging System] with a substrate like Clarity [Western ECL Substrate] with good signal duration, and then you’ll get the combination of sensitivity and resolution that you want.”
*Dr. Shao now works at the University of Southern California, where he is on the faculty of the Keck School of Medicine.
Revealing an Important Receptor in Cancers of the Gut
These western blot images, produced by Aiwen Dong, show EGFR bands in the two images acquired using the ChemiDoc Touch Imaging System (left, indicated by red boxes). Dong also includes images of the blot acquired using X-ray film (right), which was unable to detect the same band. Being able to accurately detect EGFR activity is vital for Dong, who is part of a research group developing an EGFR inhibitor as a possible therapy for cancers of the gut. Saturated bands are indicated by red highlights.
Aiwen Dong is a postdoctoral fellow at Stanford University conducting translational research on epithelial growth factor receptor (EGFR) activity. Dong and his colleagues want to understand carcinogenesis for tumors that begin in the gut, such as those involved in colorectal cancer, and are in the process of developing an EGFR inhibitor for potential therapeutic use. Performing western blot analysis for EGFR brings up the problem of detecting faint bands on a blot image; though this protein is widely expressed in epithelial cancer cells, expression levels are generally low.
Dong had been using X-ray film to image his western blots. However, as he needs to compare EGFR expression against other more abundant proteins, it’s difficult to expose for the faint EGFR band (red boxes in the blot image) while also capturing the darker bands in a usable way. When the darker bands become oversaturated, it becomes impossible to determine relative levels of protein expression.
A faint band revealed
Recently, Dong became part of the group of researchers testing the ChemiDoc Touch Imaging System. He used this instrument to image a blot membrane and detect a faint EGFR band that he could not detect using X-ray film. (Because he is involved in a patent application, he asked that other proteins on this image remain unidentified.) Dong describes how the ChemiDoc Touch Imaging System improved his ability to image a blot that compares EGFR levels to other proteins.
“Here on film I could not detect the [EGFR] band, but using the ChemiDoc Touch [Imaging System] I was able to detect it,” Dong says, pointing to his side-by-side comparison images of the same blot. “On film, I failed to see that band, even though my control was there. I also exposed the film first, so it’s not a problem of signal fading.”
Why a broad dynamic range matters
The new ChemiDoc Touch Imaging System offers an easy, flexible interface for gel and western blot imaging applications.
While getting sensitivity in detection is important, in order to get scientifically relevant data on the EGFR signaling pathway, Dong needs to be able to understand those faint bands in relation to other protein levels. “I want to compare the intensities of different bands,” says Dong. “With film, I can’t, because the stronger band always gets saturated. But with the ChemiDoc Touch [Imaging System] you can still do quantitation between bands. This is the beauty of the machine, that it can handle these differences in protein levels that film cannot handle, which shows its broad dynamic range and high sensitivity.”
In acquiring images that may have a faint band, Dong found the ChemiDoc Touch System’s preview feature especially helpful in focusing on bands of interest. “The ChemiDoc Touch System gives you a box to select your area of interest so you can let the machine decide the best exposure time to get the best dynamic range,” Dong says. By boxing key areas of the blot preview image, as Dong describes, users can optimize the exposure to best capture target protein levels.
“My PI always complains that I have saturated bands,” says Dong. “But I’m thinking now I’ll show him these new images, and he won’t be worried!”